p-Xylene (data page)
Template:Short description Template:Cleanup reorganize
Template:Use dmy dates This page provides supplementary chemical data on p-xylene.
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source and follow its directions.
Structure and properties
| Template:Chembox header | Structure and properties | |
|---|---|
| Index of refraction, nD | 1.4958 at 20 °C |
| Dielectric constant, εr | 2.2 ε0 at 20 °C |
| Surface tension[1] | 29.92 dyn/cm at 5 °C 28.27 dyn/cm at 20 °C 24.2 dyn/cm at 60 °C |
| Viscosity[2] | 0.7385 mPa·s at 10 °C 0.6475 mPa·s at 20 °C 0.5134 mPa·s at 40 °C 0.3519 mPa·s at 80 °C 0.2424 mPa·s at 130 °C |
| Solubility[3] | 0.160 g/L at 0 °C 0.181 g/L at 25 °C 0.22 g/L at 40 °C |
Thermodynamic properties
| Template:Chembox header | Phase behavior | |
|---|---|
| Triple point | 286.3 K (13.15 °C), ? Pa |
| Critical point | 617 K (344 °C), 3500 kPa |
| Std enthalpy change of fusion, ΔfusH |
17.1 kJ/mol |
| Std entropy change of fusion, ΔfusS |
59.8 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH |
35.7 kJ/mol at 138 °C |
| Std entropy change of vaporization, ΔvapS |
? J/(mol·K) |
| Template:Chembox header | Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Template:Chembox header | Liquid properties | |
| Std enthalpy change of formation, ΔfH |
−24.4 kJ/mol |
| Standard molar entropy, S |
247 J/(mol K) |
| Enthalpy of combustion, ΔcH |
−4552 kJ/mol |
| Heat capacity, cp | 181.7 J/(mol K) |
| Template:Chembox header | Gas properties | |
| Std enthalpy change of formation, ΔfH |
1.796000E+04 kJ/kg-mol[4] at 25 °C |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | 163.2 J/(mol K) at 120 °C |
| van der Waals' constants[5] | a = 3134 L2 kPa/mol2 b = 0.1809 liter per mole |
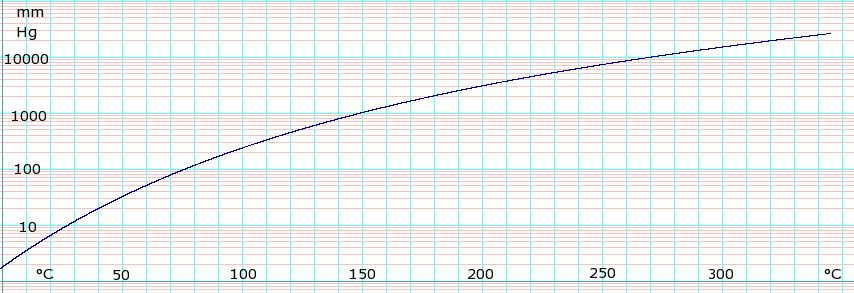
Vapor pressure of liquid
| Template:Chembox header | P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 |
| Template:Chembox header | T in °C | −8.1 | 27.3 | 54.4 | 75.9 | 115.9 | 138.3 |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
Distillation data
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Spectral data
| Template:Chembox header | UV-Vis | |
|---|---|
| Spectrum | |
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| Template:Chembox header | IR | |
| Spectrum | NIST |
| Major absorption bands | 793.94 cm−1 |
| Template:Chembox header | NMR | |
| Spectrum | AISTTemplate:Dead link |
| Proton NMR | 2.296, 7.046 |
| Carbon-13 NMR | 134.66, 128.97, 20.90 |
| Template:Chembox header | MS | |
| Spectrum | NIST |
| Masses of main fragments |
106, 91, 77 |
Template:Chemical data page general note
References
- Notes
- Bibliography